The NEOMED* NICU/Pharmacy portfolio is comprised of enteral feeding and medication filling and administration solutions.
NICU Enteral Nutrition Delivery
Turn to Avanos for solutions, including the NEOMED* brand of enteral nutrition delivery products that support the specialized needs of the neonatal and pediatric patient populations.
Designed with the NICU patient in mind, the NEOMED* enteral nutrition delivery portfolio is comprised of enteral-only feeding pumps, syringes, extension sets, and NG/OG feeding tubes and accessories. These products are available in ENFit® and non-ENFit® versions.
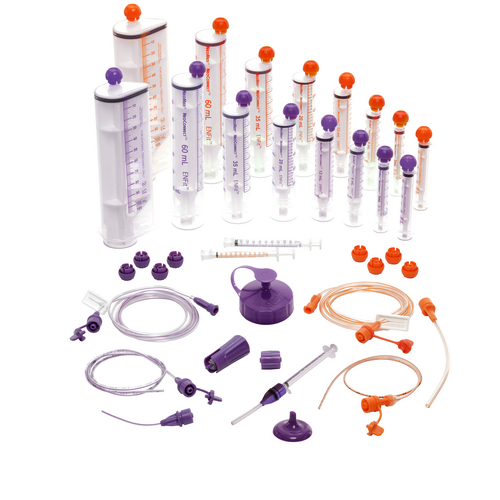

Medication Filling and Administration
FDA recommends the use of ENFit® connectors to reduce the risk of misconnection.
The NEOMED* pharmacy portfolio consists of amber and clear syringes, medication filling caps, tamper evident caps, press-in adapters, and other filling and administration accessories. Available in ENFit® and non-ENFit® versions.
NEOMED* at Home
Over the Counter, Reusable ENFit Syringes
NEOMED* at Home Syringes are available without a prescription.

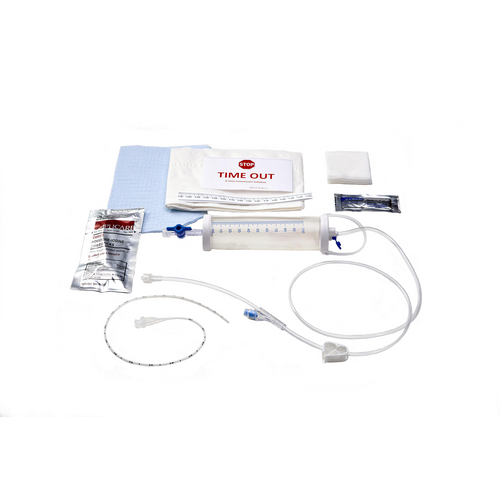
KITS & CATHETERS
Avanos offers NEOMED* Urinary Kits, Oral Care Kits, Drapes, and Lumbar Puncture Trays.
ENFit® is a trademark of GEDSA.
